Download This Sample
This sample is exclusively for KidsKonnect members!
To download this worksheet, click the button below to signup for free (it only takes a minute) and you'll be brought right back to this page to start the download!
Sign Me Up
Table of Contents
Chemistry is the study of matter, which can be described as anything that has mass and takes up space, as well as the transformations that it can go through when it is subjected to a variety of different settings and conditions.
See the fact file below for more information on Chemistry, or you can download our 31-page Chemistry worksheet pack to utilize within the classroom or home environment.
Key Facts & Information
History
- In ancient times, it was often believed that natural objects were composed of the following four fundamental elements: earth, air, fire, and water. Then, in the fourth century BCE, two Greek philosophers named Democritus and Leucippus proposed that matter did not consist of fundamental, indivisible units called atoms but rather was infinitely divisible into smaller particles. This was later developed by different chemists and scholars.
- The beginnings of what we now refer to as modern chemistry can be traced back to the 16th and 17th century. The practice of metallurgy, which involves the separation of metals from their ores, saw significant development during this period.
- The moniker “Father of Chemistry” was bestowed upon Antoine Lavoisier as a result of his numerous important contributions to the field of chemistry, including the revision and standardization of chemistry nomenclature, and the description of the properties of matter. He was a prominent French chemist and a leading figure in the chemical revolution that took place in the 18th century. He developed an experimentally based theory of the chemical reactivity of oxygen and co-authored the modern system for naming chemical substances. His work is credited with helping to establish the modern system.
- In addition to his many other contributions to science, Lavoisier is credited with developing the principle of the conservation of mass, discovering that combustion and respiration are both caused by chemical reactions with the substance that he later dubbed oxygen, and contributing to the standardization of chemical nomenclature.
Matter
- Every substance, whether it was generated by natural processes or by human ingenuity, is made up of at least one of the hundred types of atoms that have been classified as elements. Despite the fact that these atoms are made of particles that are even more fundamental, they are the crucial building blocks of chemical compounds.
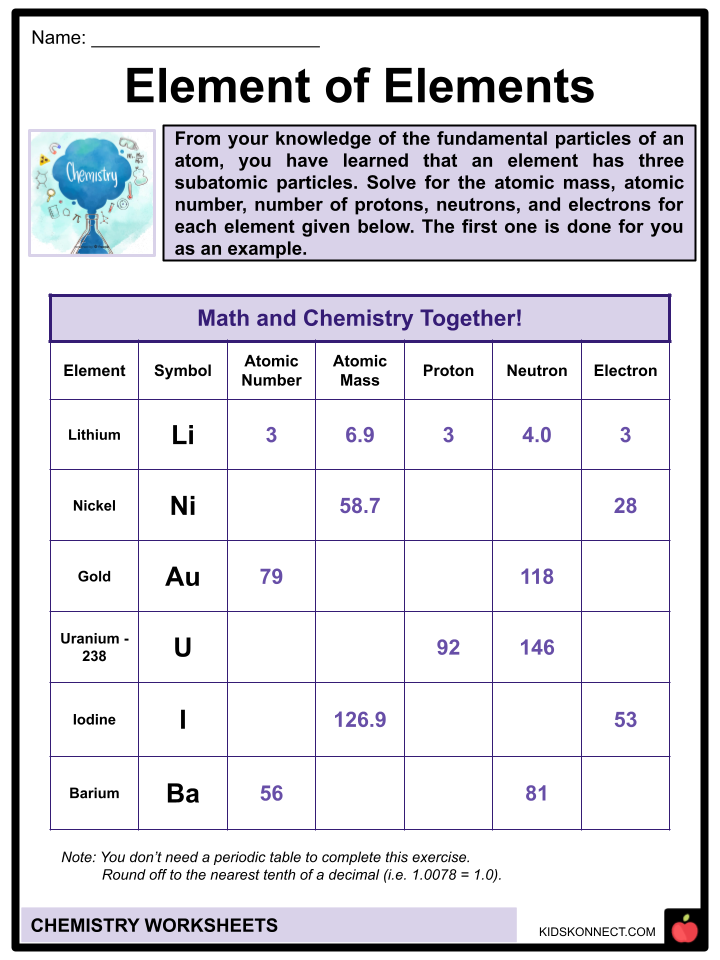
- According to general chemistry principles, atoms are made up of three fundamental particles: the proton, the neutron, and the electron.
- The proton is a subatomic particle that carries an electrical charge of one, and the number of protons in an atomic nucleus is what differentiates one kind of chemical atom from another. The neutron and the proton are similar in mass, but the neutron is chargeless while the proton is positively charged. The third constituent of an atom is called an electron, and it is smaller than that of a proton. It has a negative charge.
- Each element has a unique atomic number, which states its order in the periodic table. An element’s number of protons and electrons is equal to the atomic number, while the neutron is the difference between the atomic number and the atomic mass of each element. The sum of the number of neutrons and the atomic number makes up the element’s atomic mass.
Branches of Chemistry
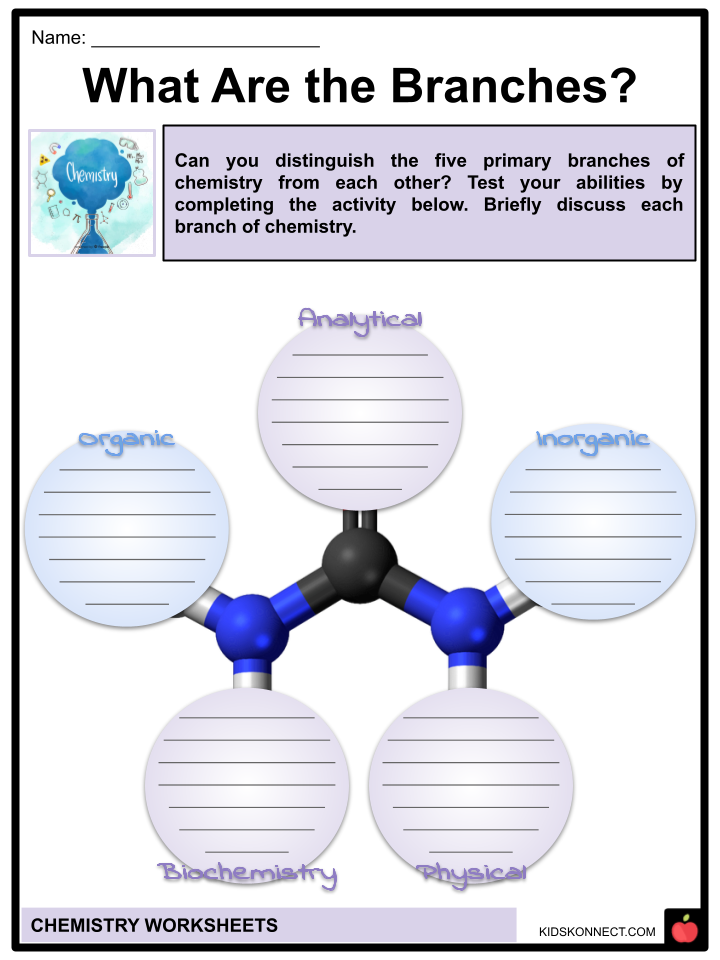
- The following are the five primary branches that makeup chemistry:
- Analytical chemistry concerns the examination of chemicals. It employs qualitative and quantitative techniques, being used in hospitals and clinics to analyze DNA, viruses, bacteria, and urine, among other things. It is also widely used in the pharmaceutical industry.
- Biochemistry is the application of chemistry principles and methods to the study of how biological systems function on a chemical level. Researchers have been able to map out the human genome, understand what different proteins perform in the body, and create solutions for a wide variety of ailments all because of biochemistry’s contributions.
- The study of the chemical substances that are found in inorganic, or non-living, materials such as minerals and metals is known as inorganic chemistry. According to the American Chemical Society (ACS), the traditional definition of inorganic chemistry is that its scope is limited to the study of substances that do not contain carbon and that this field is subsumed under organic chemistry. Inorganic chemistry is employed in the manufacture of a wide range of products, including paint, fertilizer, and sunblock.
- The study of chemical molecules containing carbon, an element regarded as necessary for maintaining life, is known as organic chemistry. This studies the composition, structure, characteristics, and reactivity of such compounds which, along with carbon, contain other non-carbon elements such as hydrogen, sulfur, and silicon. These compounds are known as “organic”. According to the American Chemical Society (ACS), organic chemistry has a wide variety of applications. Some of these applications include biotechnology, the pharmaceutical industry, the petroleum sector, and polymers.
- The study of physical chemistry draws on ideas from physics to comprehend the workings of chemistry, for instance determining how atoms travel and how they interact with one another, or discovering why some liquids, such as water, transform into vapor when subjected to high temperatures. To draw conclusions about how chemical reactions work and what gives specific materials their own distinct properties, physical chemists try to understand these phenomena on a very small scale, on the level of atoms and molecules.
Common Laboratory Equipments
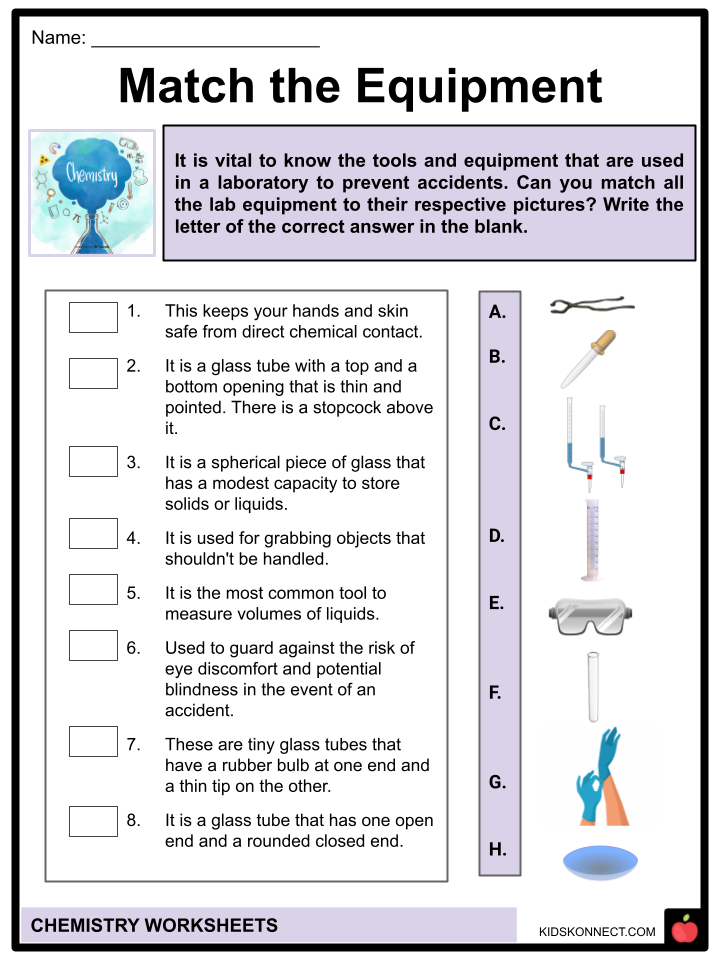
- Before anyone moves on to performing actual chemistry experiments, it is imperative that they are first taught about the most common tools and equipments found in a laboratory, their functions, usages, and safety precautions. Below are the most common laboratory equipment you must be aware of:
- All chemistry laboratories require you to wear safety goggles to guard against the risk of eye discomfort and potential blindness in the event of an accident.
- Lab aprons serve the same purpose as goggles, with the only difference being that they are intended to prevent injuries from spills and splashes rather than only protecting the eyes.
- When handling chemicals, acids, or other solutions, latex gloves come in handy to keep your hands and skin safe from direct chemical contact.
- The thin neck of the erlenmeyer flask widens as it approaches the base. This makes it simple to mix and swirl the liquid in the flask without running a significant risk of it leaking.
- A test tube is a glass tube that has one open end and a rounded closed end. Small samples are kept in test tubes.
- Ring supports, rings, and clamps are the tools used to suspend burettes, flasks, crucibles, and other heating gear above other containers or above Bunsen burners.
- A watch glass is a spherical piece of glass that has a modest capacity to store solids or liquids. In addition to serving as a beaker cover, it can be used for evaporation.
- A lab funnel is similar to any other funnel, with the exception that it was created for use in a laboratory environment.
- The most common measurement device for measuring liquid volume is a graduated cylinder.
- Droppers are tiny glass tubes with a rubber bulb at one end and a thin tip at the other. They absorb fluid, which can later be pushed out in tiny drops.
- A buret is a glass tube with a top and bottom opening that is both thin and pointed. A stopcock directly above the bottom aperture can be cranked to regulate how much liquid is emitted.
- For grabbing objects that shouldn’t be handled, use tongs or forceps. Tongs are designed specifically to hold various containers, such as test tubes and beakers.
Applications
- The use of naturally occurring compounds, as well as the production of synthetic analogs, are both aspects within chemistry. Chemical operations such as cooking, fermenting food, creating glass, and working metal have been used by humans ever since the beginning of civilization. The rewards of chemical technology can be seen in products such as vinyl, Teflon, liquid crystals, semiconductors, and superconductors in today’s world.
- Research and development
Chemists in the industry’s research and development departments use their scientific knowledge to develop new products or processes or improve existing ones. For instance, food chemists work to improve the quality of food as well as its safety, storage, and flavor; pharmaceutical chemists work to develop and analyze the quality of drugs and other medical formulations; and agricultural chemists work to develop fertilizers, insecticides, and herbicides that are necessary for large-scale crop production. - It is possible that research and development will not focus on improving the product itself but rather on the manufacturing process that is used to create the product. Chemical and process engineers
- come up with innovative ways to make the production of their goods simpler and less expensive. This could involve boosting a product’s speed and/or yield for the same amount of money, for example.
- Environmental protection
Environmental chemists are concerned with the ways in which man-made substances interact with the natural world. Their research focuses on identifying the substances and chemical reactions present in the natural processes in the earth’s soil, water, and atmosphere. To evaluate whether human activities have already contaminated the environment or will do so in the future, scientists can, for instance, collect soil, water, or air from an area of interest and then analyze it in a laboratory. - Law
Chemists can use their educational expertise to advise or campaign for scientific issues by serving in either an advisory or advocacy capacity. For instance, chemists may work in the field of intellectual property, where they may apply their scientific training to copyright issues in the scientific community. - Alternatively, chemists may work in environmental law, where they may represent special interest groups and submit paperwork to regulating agencies in order to obtain approval for certain activities. Analyses that assist law enforcement are another service that chemists can provide.
- Chemists who work in forensics examine the non-biological trace evidence that might be found at crime scenes to determine the identity of unknown compounds and compare samples to those of recognized substances. In addition, they examine narcotics and prohibited substances found at crime scenes or on individuals to identify and sometimes quantify the substances.
Chemistry Worksheets
This fantastic bundle includes everything you need to know about Chemistry across 31 in-depth pages. These ready-to-use worksheets are perfect for teaching kids about Chemistry, the study of matter and the changes it undergoes in order to form other substances and materials.



Complete List of Included Worksheets
Below is a list of all the worksheets included in this document.
- Chemistry Facts
- Match the Equipment
- Fill in the Blanks
- Little Alchemist
- Came From Chemistry?
- Know Thy Father
- Element of Elements
- Zoom in on an Atom
- Chemist Cook
- What Are the Branches?
- My Takeaways
Frequently Asked Questions
What is Chemistry?
Chemistry is the scientific study of matter and its properties, composition, and behavior. It is the branch of science that deals with the study of the properties of substances and the ways in which they interact with each other and with energy. It also includes the study of chemical reactions and the manipulation of matter at the atomic and molecular level.
What are some of the main branches of chemistry?
Some of the main branches of chemistry include inorganic chemistry, which deals with the properties and behavior of non-organic compounds; organic chemistry, which deals with the properties and behavior of organic compounds; biochemistry, which deals with the chemical processes that occur in living organisms; analytical chemistry, which deals with the identification and measurement of the composition of substances; and physical chemistry, which deals with the mathematical and theoretical aspects of chemistry.
How do chemical reactions occur?
Chemical reactions occur when the atoms in a substance are rearranged to form new compounds. This can happen through a variety of mechanisms such as the transfer of electrons, the formation of new bonds between atoms, or the breaking of existing bonds. These changes in the arrangement of atoms result in the formation of new substances with different properties than the original reactants.
Link/cite this page
If you reference any of the content on this page on your own website, please use the code below to cite this page as the original source.
Link will appear as Chemistry Facts & Worksheets: https://kidskonnect.com - KidsKonnect, January 12, 2023
Use With Any Curriculum
These worksheets have been specifically designed for use with any international curriculum. You can use these worksheets as-is, or edit them using Google Slides to make them more specific to your own student ability levels and curriculum standards.



