Download This Sample
This sample is exclusively for KidsKonnect members!
To download this worksheet, click the button below to signup for free (it only takes a minute) and you'll be brought right back to this page to start the download!
Sign Me Up
Table of Contents
An atom is made up of one positively charged nucleus and several negatively charged electrons. When atoms are near to one another, their electron clouds and nuclei come into contact. If this contact reduces the overall energy of the system, the atoms link together to create a molecule.
See the fact file below for more information on Molecules or alternatively, you can download our 22-page Molecules worksheet pack to utilize within the classroom or home environment.
Key Facts & Information
HISTORY OF MOLECULES
- Numerous ancient civilizations believed that life and all existence were formed out of the basic components of nature, i.e., earth, air, wind, water, and fire, which are the five elements.
- However, throughout time, many philosophers came to propose that all things were made up of small, inseparable objects that could not be created or destroyed.
- Molecular theory and atomic theory have always been related.
- The concept that matter is made up of “discrete units” was originally mentioned in ancient India around 500 B.C, where followers of Jainism held that all things were made up of little indivisible parts that merged to make more advanced forms.
- However, this was essentially a philosophical idea, and it was not until the development of atomic theory and modern chemistry from the 18th century that scientists began to propose that particles, when combined, created the fundamental building blocks of all things.
- They termed them “molecules,” after the Latin word for “mass” or “barrier.” However, in contemporary particle theory, the word refers to tiny units of mass.
- Atoms and molecules were not included in the realm of hard science until the late 18th and early 19th centuries, thanks to Antoine Lavoisier’s Law of Conservation of Mass and John Dalton’s Law of Multiple Proportions.
- The former suggested that elements are basic substances that cannot be dissolved further, while the latter proposed that each element consists of a single, unique sort of atom that may be combined to produce chemical compounds.
CHARACTERISTICS OF MOLECULES
- A molecule, according to its proper definition, is the smallest particle of a material that preserves its chemical and physical characteristics.
- They are made up of two or more atoms, or a collection of similar or dissimilar atoms bonded together by chemical forces.
- The molecular mass is the size of a given molecule.
- Molecules are separated from one another and move randomly all the time.
- The tiniest component of a substance, an atom, cannot exist alone. In contrast, a molecule may be thought of as the smallest component of matter. An element or component of a compound that may live alone or freely in the natural condition
- It can be made up of atoms of a single chemical element, such as oxygen (O2), or of several elements, such as water (H2O).
- As matter components, molecules are common in organic substances (and hence biology) and are responsible for life-giving materials such as liquid water and breathable atmospheres.
THE PHYSICAL STATE OF A MOLECULE
- The physical state and characteristics of a molecule are heavily influenced by the sort of chemical bonding it exhibits. Because of distinct sorts of intermolecular attractions, such as different types of polar interactions, molecular compounds, also known as covalent compounds, exhibit a wide range of physical characteristics.
- When compared to ionic compounds, the melting and boiling temperatures of molecular compounds are often low. This is because the energy required to break the ionic bonds in a crystalline ionic compound is significantly less than that necessary to disrupt the intermolecular interactions between molecules.
- Because molecular compounds are made up of neutral molecules, their electrical conductivity is often low, whether in solid or liquid form. Because of their rigid structure, ionic compounds do not conduct electricity when solid, but they do when melted or dissolved into a solution.
- The water solubility of molecular compounds varies and is mostly determined by the sort of intermolecular interactions involved. Water is often soluble in substances that show hydrogen bonding or dipole-dipole interactions, but substances that exhibit just London dispersion forces are generally insoluble.
- Most ionic chemicals, although not all, are very soluble in water.
- In comparison to ionic compounds, covalent compounds are softer, have lower boiling and melting temperatures, are more combustible, are less soluble in water, and do not conduct electricity.
- Molecular polarity influences the melting and boiling temperatures, solubility, and other physical characteristics of molecular compounds.
THREE TYPES OF MOLECULES
- These atoms are kept together by chemical forces or chemical connections.
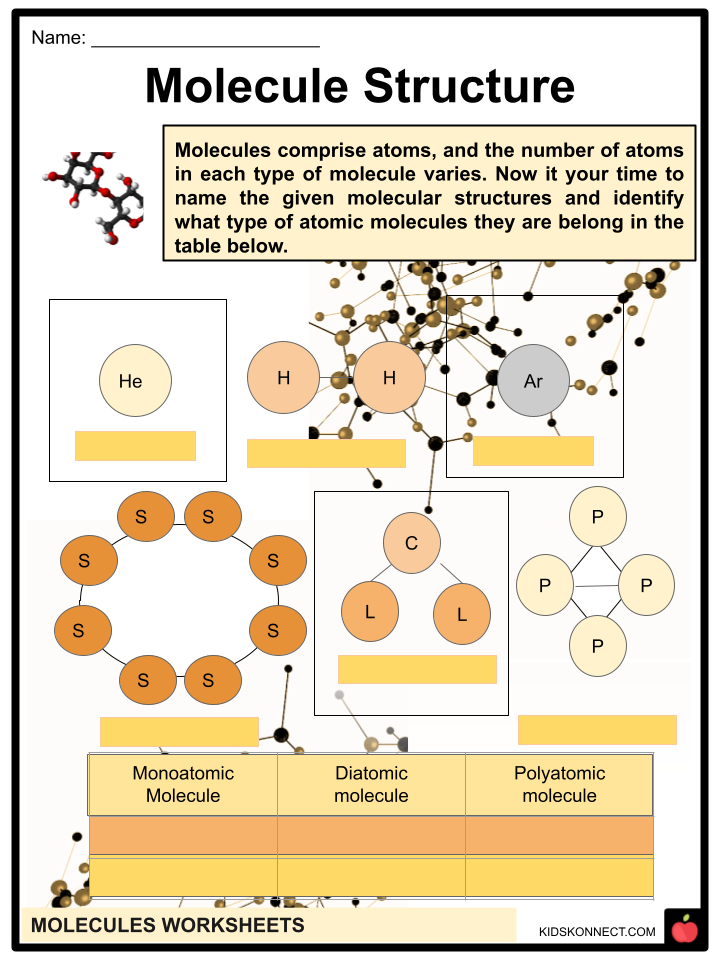
- The element molecule is the most fundamentally pure form of matter that cannot be chemically disassembled into a less complex form. The molecules of the element are monatomic, consisting of a single solid atom.
Compound Molecules
- Compound molecules are made up of atoms from two or more distinct elements together in a consistent weight ratio. It is possible to decompose it chemically into a simpler form of matter.
- Mixture molecules are made up of two or more different elements or compounds that have been physically mixed together. It may be physically divided into its constituent parts, and it frequently retains a lot of the characteristics of those parts.
- Monatomic, which stands for “one atom,” is a combination of the terms “mono” and “atomic.” It refers to gases as monatomic gases and is used in both physics and chemistry.
- All the chemical elements are monatomic gases when they are in the gaseous phase at high enough temperatures.
- A single-atom, covalent-bond-free molecule is referred to as a monatomic. Noble gases are monatomic gases because of their natural inertness, which is a characteristic of these gases.
- Helium, Neon, Argon, and Krypton are some examples of noble gases, which are monatomic (figure 6).
Diatomic Molecules
- Diatomic molecules are those composed of two atoms. Homonuclear diatomic molecules are made of only one element, while heteronuclear molecules are made of two distinct elements.
- Diatomic substances are pure forms of non-metals. O2, Cl2, and Br2 are a few examples of molecules of an element. (figures 7 and 8).
- A stable system (bound state) with three or more atoms characterizes polyatomic molecules.
- A molecular ion made up of two or more atoms bound by covalent bonds is referred to as a polyatomic ion. It’s also known as a radical.
- Molecules constantly oscillate through vibrational and rotational movements about given equilibrium geometries, or bond lengths and angles.
- Different molecules are distinguished from one another by the precise number of atoms in their molecular formula. Examples of polyatomic compounds are phosphorus (P4) (figure 9) and sulfur (S8).
Macromolecules
- Macromolecules are huge molecules made up of thousands of covalently bonded atoms. Carbohydrates, lipids, proteins, and nucleic acids are all macromolecules.
- Macromolecules are generated when numerous monomers bind together to form a polymer. Carbohydrates are made up of carbon, oxygen, and hydrogen.
The valence shell electron pair repulsion (VSEPR) theory
- Covalent molecules, unlike ionic compounds, which have extended crystal lattices, are distinct entities with unique three-dimensional structures.
- The form of a molecule is governed by the fact that negatively charged electrons in covalent bonds repel one another. The valence shell electron pair repulsion (VSEPR) theory describes this notion.
- For example, in BeCl2, the two covalent bonds stay as far apart as possible, eventually ending up 180° apart. As a result, a linear molecule is formed (figure 1).
- The three covalent bonds in BF3 oppose each other, forming 120° angles in a plane, giving rise to the trigonal planar shape (figure 3).
- The four covalent bonds in CCl4 form a three-dimensional arrangement, pointing toward the corner of a tetrahedron and forming bond angles of 109.5°. The shape of CCl4 is considered to be tetrahedral (figure 4).
- Polar molecules are formed when the electronegativity of the atoms in the molecule differs.
- Dipoles that are completely contradictory cancel each other out.
Molecular Bond
- Molecules generate two kinds of bonds: ionic bonds and covalent bonds. Ionic bonds convey electrons from one atom to another, whereas covalent bonds exchange electrons.
- The energy change involved with ionic bonding is determined by three processes: ionization of an electron from one atom, acceptance of the electron by the second atom, and Coulomb attraction of the resultant ions.
- Space-symmetric wave functions are involved in covalent bonding. To interact with other molecules, atoms need a linear combination of wave functions (hybridization).
- Many distinct types of chemical bonds combine to generate chemical units. When an electron moves from one atom to another, an ionic connection is formed. When two or more atoms share electrons, they form a covalent connection.
- Van der Waals bonds form as a result of the attraction of charge-polarized molecules and are much weaker than ionic or covalent interactions.
- The attraction of two electrically polarized molecules results in the formation of a der Waals bond.
- The van der Waals force aids in the formation of a secondary bond between polymer molecules.
- Polyethylene, for example, contains a polymer chain composed of repeated monomers of two covalently bound carbon atoms each containing two hydrogen atoms apiece.
- The van der Waals forces hold the polymer chains of polyethylene together.
Molecules Cycle Worksheets
This is a fantastic bundle that includes everything you need to know about Molecules across 22 in-depth pages. These are ready-to-use worksheets that are perfect for teaching about Molecules, which are what result when atoms link together.

Complete List of Included Worksheets
Below is a list of all the worksheets included in this document.
- Molecules Facts
- Fact or Bluff
- Word Search
- Remember Me
- Molecule Structure
- What Am I?
- Build Me Up
- Molecular Bond
- VSEPR Theory
- What Best Describe Me?
- Think Outside The Box
Frequently Asked Questions
What is the definition of a molecule?
All molecules are made up of one or more atoms connected by chemical bonds, and they form the basis of chemistry. If a molecule contains more than one atom, the atoms can be the same or different, with some biological molecules being made up of thousands of atoms.
Are there molecules in the body?
There are thousands of different molecules in the human body, and they all serve critical tasks. The four molecules of life are proteins, lipids, carbohydrates, and nucleic acids. Each of these four groups is vital for every single organism to maintain life.
What is the difference between a molecule and a cell?
A cell is made of molecules, and a molecule is made up of atoms. Simply put, a cell is made up of macromolecules, such as proteins, lipids, etc., and a molecule is a particular configuration of atoms.
Link/cite this page
If you reference any of the content on this page on your own website, please use the code below to cite this page as the original source.
Link will appear as Molecules Facts & Worksheets: https://kidskonnect.com - KidsKonnect, July 27, 2022
Use With Any Curriculum
These worksheets have been specifically designed for use with any international curriculum. You can use these worksheets as-is, or edit them using Google Slides to make them more specific to your own student ability levels and curriculum standards.


