Download This Sample
This sample is exclusively for KidsKonnect members!
To download this worksheet, click the button below to signup for free (it only takes a minute) and you'll be brought right back to this page to start the download!
Sign Me Up
Table of Contents
A negatively charged subatomic particle is an electron. It is one of the fundamental particles that make up matter. Electrons are found in atoms and play a crucial role in chemical reactions and electrical conductivity.
See the fact file below for more information on Electrons, or you can download our 24-page Electron worksheet pack to utilize within the classroom or home environment.
Key Facts & Information
OVERVIEW OF ELECTRON IN QUANTUM MECHANICS
- According to quantum mechanics, all electrons are stated to have the identical mass, charge, and rotational spin. As a result, electrons are able to switch positions with other electrons without much change.
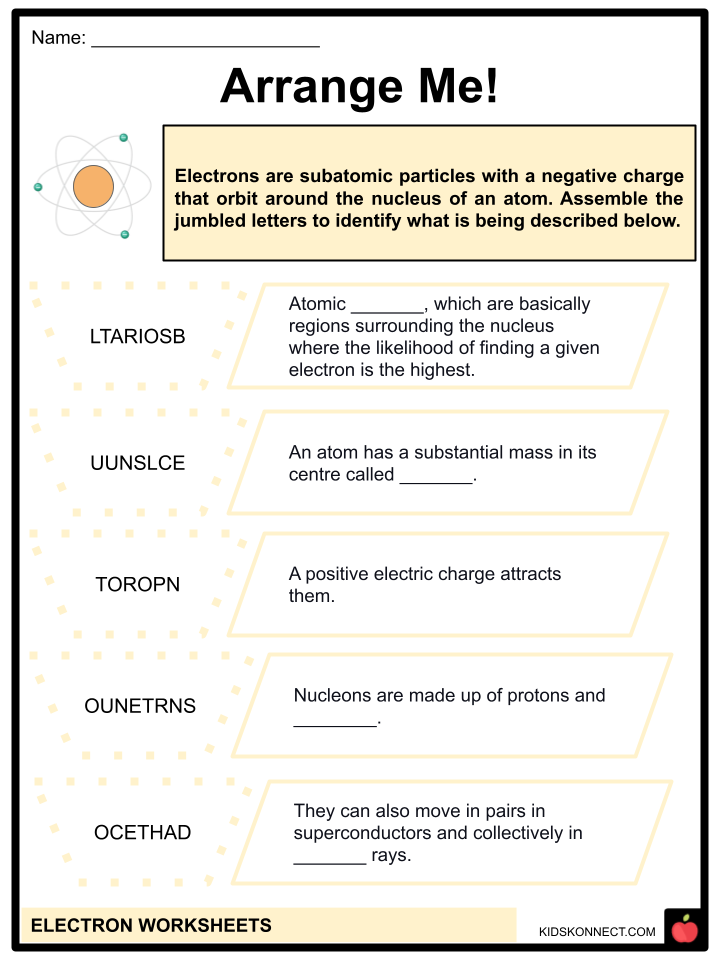
- Due to their same negative electric charge, electrons repel one another. Yet, protons with a positive electric charge attract them. An atom is made up of protons, neutrons, and electrons, which are subatomic particles. Anything will be negatively charged if it contains more electrons than protons. If a thing has more protons than electrons, it might become positively charged.
ELECTRON SIZE AND MASS
- The electron is so small that it cannot be disassembled any further. In fact, it is so little its actual size has not been identified. When anything gets shrunk down to the size of an electron, it is impossible to measure it since the edges cannot be located.
- Yet, according to scientific estimates, the radius is roughly 2.82 x 10-15 cm.
- That indicates that nearly 5,640,000,000,000 electrons might be lined up in one mm; a lot of electrons!
- An electron weighs 9.10938356 kilograms or 10-31 coulomb. In comparison to the mass of the proton, the mass of the electron is small.
- Because of their small size and mass, electrons provide more interesting study subjects for quantum mechanics than for classical mechanics.
- This is due to the distinct behavior of matter at the quantum level. Heisenberg’s uncertainty principle, for instance, states that an electron’s position and velocity are subject to significantly greater uncertainty than those of a proton or a neutron.
- Atomic orbitals, which are basically regions surrounding the nucleus where the likelihood of finding a given electron is the highest, are where electrons are distributed around the nuclei of atoms.
SIGNIFICANCE OF ELECTRON
- Electrons are essential because they give atoms, which are the building blocks of all things, structure, and volume. The combining of individual atoms into pairs or groups by means of chemical bonds involves electrons.
- Without electrons, there would be no bonding force to hold the atoms together.
- As a result, atoms would not be able to interact in the different ways we see them doing so today, and life as we know it would not exist.
- Major advancements were made in the study of gravity, electromagnetic, thermal conductivity, and electricity as a result of Joseph Thomson’s discovery of electrons.
- Thomson was duly awarded the Nobel Prize in Physics in 1906 for his contributions. Yet, other scientists, including Richard Laming and Richard Fleming, had predicted the potential existence of electrons prior to Thomson’s statement and discovery of their existence.
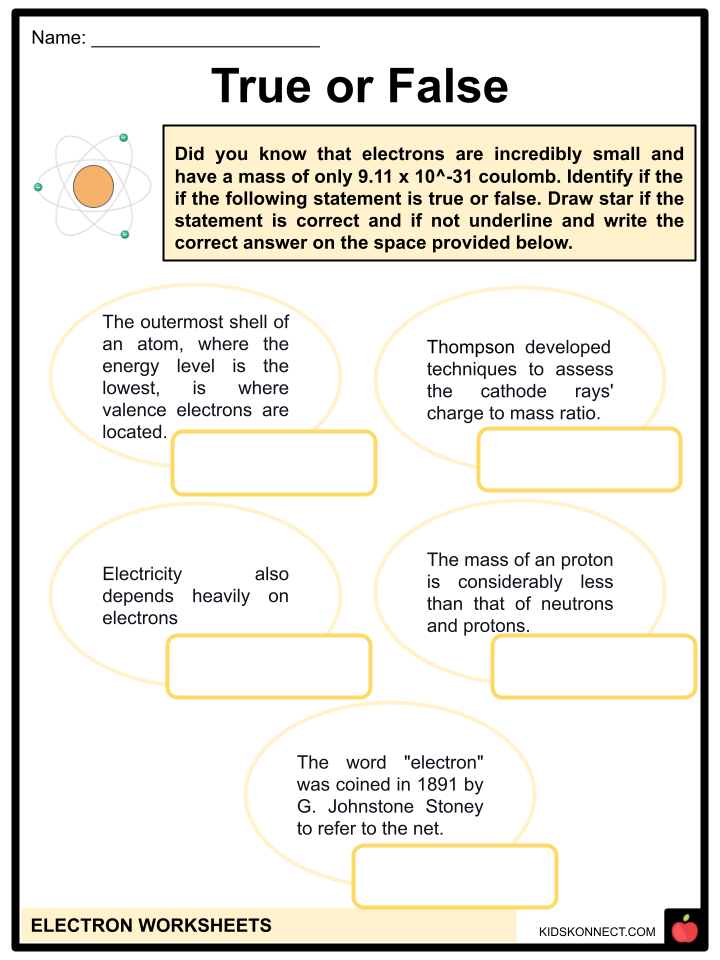
- Electricity also depends heavily on electrons.
- When electrons move from a negatively charged end to a positively charged end, they produce flow or current, which results in electricity.
THE NATURE AND PROPERTIES OF ELECTRON
- Electrons are thought to be dual in nature and have diverse features.
- The French physicist Louis de Broglie claimed that electrons could exist as a particle and a wave.
- American physicists Clinton Davisson and Lester Germer later verified this by doing tests on electron diffractions to get this result.
- Protons are positively charged particles, while electron is negatively charged. The mass of an electron is considerably less than that of neutrons and protons.
- An electron’s mass is 1836 times greater than a proton’s. Each electron carries 1.602 x 10-19 coulombs of negative charge.
- Because each electron has the same mass, electrical charge, and rotating spin, they can freely move and alternate locations. They are the primary electrical transporters in solids.
- Electrons are generally found in atoms, however, they can exist independently of an atom as well.
- They can also move in pairs in superconductors and collectively in cathode rays. Electricity is produced when electrons flow and create current.
- The atomic theory states that electrons form shells around the atomic nucleus. It was also possible to find it wherever in an atom.
- Chemical bonds are produced when atoms exchange or transfer electrons.
COMPOUND AND ELECTRON
- Afterward, protons, electrons, and neutrons—the fundamental subatomic particles—were identified by scientists. An atom has a substantial mass in its center, called a nucleus.
- Protons and neutrons are found in the nucleus. It has been noted that the sun is in the middle of the solar system, and the planets orbit it.
- Similar to this, the nucleus is in the center of an atom, and the electrons orbit it. J. J. Thompson deserves the credit for discovering the electron.
- In an experiment he conducted with cathode rays, he demonstrated that they are charged particles. Cathode rays’ velocity was considerably lower than the speed of light. He developed techniques to assess the cathode rays’ charge-to-mass ratio.
- Thompson came to the conclusion that the hydrogen ion’s mass is 1/1000th that of the cathode rays (which is a proton). Nowadays, electrons are the name given to these negatively charged cathode ray particles.
- Electrons are incredibly light, negatively charged particles. The total of an atom’s protons and neutrons determines its mass. In contrast to neutrons, which have no charge, protons are positively charged particles. Since there are an equal number of protons and electrons, an atom is electrically neutral. Protons and neutrons make up nucleons.
ELECTRON TYPES & EXAMPLES
- Electrons have been split into two types by scientists. These two types of electrons are known as valence electrons and core electrons.
- The outermost shell of an atom, where the energy level is the highest, is where valence electrons are located. The lowest energy level of an atom’s innermost shell is where core electrons are located.
- These are subatomic particles that can exist as free electrons or travel about on the exterior of atomic nuclei.
- These electrons or tiny particles are so important that they are why we can see, feel, and hear everything around us.
- The movement of electrons via a conductor, such as a wire, produces electricity. This is how we power our homes, schools, and devices. Without electrons, we couldn’t turn on the lights, charge our phones, or watch TV.
- Magnets are made up of atoms that have their electrons aligned in a specific way. This alignment creates a magnetic field, which can attract or repel other magnets or certain metals. Kids may have played with magnets with interest or seen them used to hold papers on a fridge or in science class.
- Lastly, when we touch something, electrons from our skin interact with electrons in the object we’re touching. This interaction creates a force that we feel as touch or pressure. So, when we think of something soft or hard, it’s because of the interaction between electrons.
Electron Worksheets
This fantastic bundle includes everything you need to know about Electrons across 24 in-depth pages. These are ready-to-use worksheets that are perfect for teaching kids about Electrons. Electrons are found in atoms and play a crucial role in chemical reactions and electrical conductivity.




Complete List of Included Worksheets
Below is a list of all the worksheets included in this document.
- Electron Facts
- Arrange Me
- True or False
- Key Scientist
- Answer Me
- Compare Us
- Draw My Importance
- Electron Carrier
- Don’t Be Shy on Camera
- Watch and Learn
- Man Made Structure
Frequently Asked Questions
What is an electron?
An electron is a subatomic particle that carries a negative charge and orbits around the nucleus of an atom. Electrons are one of the building blocks of matter and play a critical role in chemical reactions and electricity.
What is the mass of an electron?
The mass of an electron is approximately 9.11 x 10^-31 kilograms, which is about 1/1836th the mass of a proton. Electrons are much lighter than protons and neutrons, the other two subatomic particles that make up an atom.
How are electrons arranged in an atom?
Electrons are arranged in shells or energy levels around the nucleus of an atom. The electrons in the first shell, closest to the nucleus, have the lowest energy level. The electrons in the outermost shell, or valence shell, have the highest energy level and are involved in chemical reactions.
What is an electron’s charge?
An electron has a negative charge of -1. This means that electrons are attracted to positively charged particles, such as protons, and repel other negatively charged particles, such as other electrons.
What is electron configuration?
Electron configuration refers to the arrangement of electrons in an atom. The configuration of electrons in an atom is determined by the number of electrons in each energy level or shell. The electron configuration of an atom can help predict the chemical properties of that element.
Link/cite this page
If you reference any of the content on this page on your own website, please use the code below to cite this page as the original source.
Link will appear as Electron Facts & Worksheets: https://kidskonnect.com - KidsKonnect, March 30, 2023
Use With Any Curriculum
These worksheets have been specifically designed for use with any international curriculum. You can use these worksheets as-is, or edit them using Google Slides to make them more specific to your own student ability levels and curriculum standards.



