Download This Sample
This sample is exclusively for KidsKonnect members!
To download this worksheet, click the button below to signup for free (it only takes a minute) and you'll be brought right back to this page to start the download!
Sign Me Up
Table of Contents
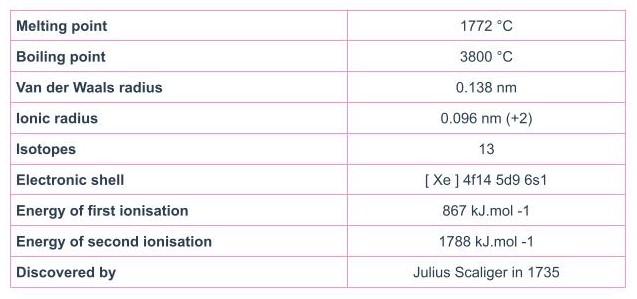
Platinum is a periodic table element with the atomic number 78 and the atomic mass 195. It is a very uncommon element found in the Earth’s crust. Platinum is a dense, lustrous, and precious metal known for its remarkable corrosion resistance, making it highly valued for various industrial and jewelry applications. Platinum’s exceptional properties and scarcity make it one of the most sought-after and valuable precious metals.
See the fact file below for more information about Platinum, or you can download our 27-page Platinum worksheet pack to utilize within the classroom or home environment.
Key Facts & Information
HISTORY
- Because of its grey-white silvery color, platinum is derived from Platina (Spanish for ‘small silver’). It is one of the rarest elements and one of the most precious due to its 5 g/kg availability in the Earth’s crust.
- Although European scientists discovered it relatively late, it has been detected in Egyptian tombs dating back to 1200 BC.
- The oldest known worked platinum specimen is from a 7th-century BCE Egyptian casket discovered in Thebes and dedicated to Queen Shapenapit.
- Otherwise, this metal was unknown in Europe and Asia for the following two millennia.
- However, some individuals could work platinum on South America’s Pacific coast, as evidenced by burial items dating back 2000 years.
- In 1557, the Italian scholar Julius Scaliger first documented a unique metal from Spanish Central America, which defied melting and was identified as platinum.
- Later, in 1735, Antonio Ulloa encountered this unusual metal, but an encounter with the Royal Navy during his return to Europe led him to London.
- Members of the Royal Society showed keen interest in learning about this new metal, and by the 1750s, discussions and reports about platinum had spread across Europe.
ABOUT
- Platinum is a silver-white metallic element found worldwide, frequently combined with other metals like iridium, osmium, or nickel.
- It is malleable and ductile, does not oxidize in air, and is employed as a catalyst in electrical components, jewelry, dentistry, and electroplating.
- Platinum is as resistant to corrosion and tarnishing as gold and will not oxidize in the air, no matter how hot.
- Platinum is capable of conducting electricity. Platinum and rhodium, ruthenium, palladium, osmium, and iridium constitute the platinum group metals (PGM).
- This element has the Atomic Number 78 and the Element Symbol Pt.
- Elements, such as gas, solid, or liquid, are categorized according to their physical states (States of Matter).
- Platinum is a solid element and is classified as a “Transition Metal” and is found in Periodic Table Groups 3 – 12.
- Elements classed as Transition Metals are ductile, malleable, and capable of conducting electricity and heat.
- Metals account for over 75% of all elements in the Periodic Table, as stated in the List of Metals.
USES AND PROPERTIES
- Appearance: A gleaming, silvery-white metal that is as corrosion-resistant as gold.
- Uses: Platinum is commonly used in jewelry. Its principal use, however, is in catalytic cars, trucks, and bus converters.
- This accounts for roughly half of the annual demand. Platinum is particularly effective at transforming engine emissions into less hazardous waste products.
- It is used for electrodes in laboratories.
- Platinum is used in the chemical industry as a catalyst to produce silicone, nitric acid, and benzene. It is also used as a catalyst to increase the efficiency of fuel cells.
- Platinum is used in the electronics sector for computer hard disks and thermocouples.
- Platinum also produces optical fibers and LCDs, spark plugs, pacemakers, turbine blades, and dental fillings.
- Platinum compounds are chemotherapeutic medications that are used to treat cancer.
- To make their timepieces more unique, some watchmakers employ platinum.
- Natural Abundance: Alluvial deposits contain uncombined platinum. Most commercially produced platinum comes from South Africa, where the mineral cooperite (platinum sulfide) is mined.
- Some platinum is produced as a byproduct of the processing of copper and nickel.
EFFECTS OF PLATINUM
- Platinum is a precious metal. Platinum concentrations in soil, water, and air are deficient. Platinum deposits can be discovered in several regions, primarily South Africa, the Soviet Union, and the United States.
- Platinum is utilized as a component in various metal products, such as electrodes, and it can act as a catalyst in various chemical reactions.
- Platinum bonds are frequently used as a cancer treatment. The health effects of platinum are greatly reliant on the type of connections formed, as well as the person’s exposure level and immunity.
- Platinum as a metal is not very hazardous, but platinum salts can have several adverse health effects, including:
- Platinum can potentially increase the toxicity of other harmful substances in the human body, such as selenium.
- The usage of platinum in metal goods is not known to generate many environmental issues, but it is known to cause significant health concerns in the workplace.
- Platinum is released into the atmosphere via the exhausts of cars that run on leaded gasoline. Consequently, specific locations, such as garages, tunnels, and trucking company facilities, might exhibit elevated levels of platinum in the air.
- The impacts of platinum on animals and the environment have yet to be thoroughly investigated. The only thing we know is that following uptake, platinum will accumulate in plant roots.
- It is unclear whether ingesting platinum-containing plant roots can harm animals or humans.
- In soil, there is a possibility that microorganisms can transform platinum compounds into potentially more hazardous substances, although our understanding of this process remains quite limited.
FUN FACTS
- Platinum is one of the rarest elements on Earth. All the platinum ever mined was estimated to fit into an average-sized living room.
- Platinum is highly resistant to corrosion and tarnish, which makes it ideal for use in jewelry and various industrial applications, including spacecraft.
- Platinum has an exceptionally high melting point of 3,215.1 degrees Fahrenheit (1,768.4 degrees Celsius), making it one of the most heat-resistant elements.
- Platinum is often used in high-end jewelry because of its purity and ability to maintain its luster over time. It’s also hypoallergenic, making it an excellent choice for sensitive skin.
- Platinum is a critical element in catalytic converters used in vehicles to reduce harmful emissions, helping to reduce air pollution.
- Platinum is found in meteorites and is believed to be a component of the Earth’s core. It’s also used in space exploration due to its resistance to extreme temperatures and radiation.
- Some countries, such as the Russian ruble, have used platinum in their currency. Additionally, platinum is used for prestigious awards, like the Grammy and Emmy awards.
- Compounds containing platinum, such as cisplatin, are used in cancer treatment to disrupt the growth of cancer cells.
- An album going “platinum” refers to its sales success in the music industry. This term originally referred to the platinum vinyl records used for high-sales awards.
Platinum Worksheets
This fantastic bundle includes everything you need to know about Platinum across 27 in-depth pages. These ready-to-use worksheets are perfect for teaching kids about Platinum. Platinum is a dense, lustrous, and precious metal known for its remarkable corrosion resistance, making it highly valued for various industrial and jewelry applications. Platinum’s exceptional properties and scarcity make it one of the most sought-after and valuable precious metals.





Complete List of Included Worksheets
Below is a list of all the worksheets included in this document.
- Platinum Facts
- True or False
- 4 Pics 1 Word
- Element Profile
- Searching for…
- Nanotechnology
- Platinum Catalysts
- Made in Platinum
- PLATINUM
- Jewelry Metals
- Crafting with Platinum
Frequently Asked Questions
What is platinum?
Platinum is a chemical element with the symbol Pt and atomic number 78. It is a dense, silvery-white metal that belongs to the platinum group of elements in the periodic table, which also includes palladium, rhodium, ruthenium, iridium, and osmium.
Where is platinum found?
Platinum is relatively rare in the Earth’s crust and is often found in association with other precious metals. The majority of platinum production comes from mines in South Africa, Russia, and Zimbabwe. It can also be found in smaller quantities in other regions around the world, including North and South America.
What are the primary uses of platinum?
Platinum has numerous industrial and commercial applications due to its remarkable properties. Some common uses include:
- Catalytic converters in vehicles to reduce harmful emissions.
- Jewelry and adornments, often in the form of platinum rings, necklaces, and earrings.
- In the production of laboratory equipment and electrical contacts due to its resistance to corrosion.
- As a catalyst in various chemical processes, including the production of nitric acid and in the petroleum industry.
What is the price of platinum?
The price of platinum can vary significantly over time due to factors like supply and demand, economic conditions, and geopolitical events. Historically, platinum has been more expensive than gold, but its price is influenced by various factors. As of my knowledge cutoff date in early 2022, platinum was trading at around $1,000 to $1,200 per troy ounce. However, prices can fluctuate, and it’s essential to check the current market value for the most up-to-date information.
Is platinum a precious metal like gold and silver?
Yes, platinum is considered a precious metal, along with gold and silver. Precious metals are valued for their rarity, beauty, and various industrial applications. Platinum, in particular, is highly regarded for its durability and resistance to corrosion, making it a popular choice for both jewelry and industrial uses. It is also used as a store of value, similar to gold and silver, and is often viewed as a safe haven investment during times of economic uncertainty.
Link/cite this page
If you reference any of the content on this page on your own website, please use the code below to cite this page as the original source.
Link will appear as Platinum Facts & Worksheets: https://kidskonnect.com - KidsKonnect, November 9, 2023
Use With Any Curriculum
These worksheets have been specifically designed for use with any international curriculum. You can use these worksheets as-is, or edit them using Google Slides to make them more specific to your own student ability levels and curriculum standards.