Download This Sample
This sample is exclusively for KidsKonnect members!
To download this worksheet, click the button below to signup for free (it only takes a minute) and you'll be brought right back to this page to start the download!
Sign Me Up
Table of Contents
Argon is a noble gas considered the third-most abundant gas in Earth’s atmosphere. It is the most abundant element in the Earth’s crust. It is also the most abundant noble gas. It is found in group 18 of the periodic table of elements, along with helium, neon, argon, krypton, xenon, radon, and oganesson. It is a colorless, odorless, and tasteless gas that remains without color in its solid and liquid form. One of its industrial applications is as an effective electrical insulator and component of light bulbs.
See the fact file below for more information about Argon, or you can download our 25-page Argon worksheet pack to utilize within the classroom or home environment.
Key Facts & Information
ETYMOLOGY
- Argon’s name was derived from the Greek word “argos” meaning “lazy,” “idle” or “inactive” — a reference to how non-reactive it is and how it forms very few compounds.
- Argon is a noble gas.
- The term “noble gas” was coined by German chemist Hugo Erdmann in 1898.
- It had been derived from “noble metals,” a term used to refer to nonreactive metals.
HISTORY
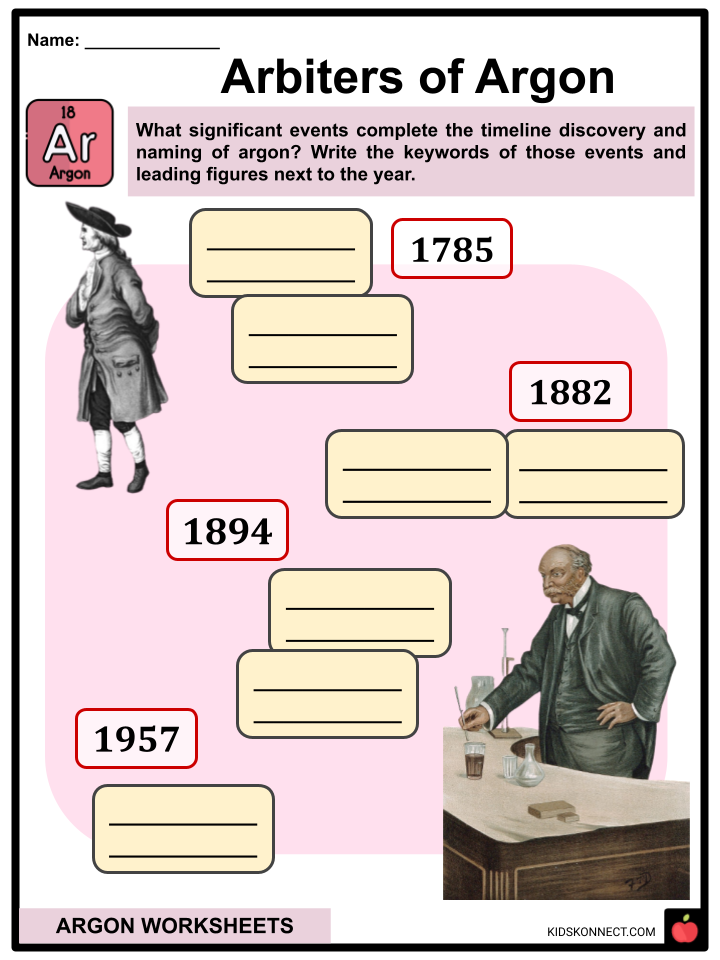
- The discovery of argon can be traced to 1785 when English chemist Henry Cavendish became the first scientist to demonstrate that air has different gas components than nitrogen, oxygen, and carbon dioxide.
- In his experiments, Cavendish examined samples of clean air.
- Cavendish argued that there was a non-reactive gas in the air, but he was not able to isolate which particular element it was.
- The next documented encounter of argon was in 1882 when H.F. Newall and W.N. Hartley conducted independent research and observed new spectral lines in the emission of air that was not assigned to any known element.
- In 1894, argon was isolated from air by English scientist Lord Rayleigh and Scottish chemist Sir William Ramsay by replicating Cavendish’s experiment.
- Rayleigh and Ramsay were able to remove oxygen, nitrogen, carbon dioxide, and water from a sample of clean air.
- They discovered argon and other noble gases present in the air residue, for which they received the Nobel Prize in Chemistry and Physics in 1904.
- These elements, however, accounted for very little of the sample’s total mass.
- A was argon’s initial symbol in the periodic table of elements.
- It was changed to Ar by the International Union of Pure and Applied Chemistry (IUPAC) in 1957.
ELEMENT QUICK FACTS
- In the periodic table, argon is represented by the symbol Ar and the atomic number 18.
- Argon has 18 protons, 18 electrons, and 22 neutrons.
- Its outermost (valence) shell is full of eight electrons,
which means it is a highly stable element. - It is part of group 18, period 3, and p-block.
- The atomic weight of argon is 39.948.
- Its density is 1.784 g/L at standard temperature and pressure.
- Its boiling point is -185.85 °C, while its melting point is -189.35 °C.
- Argon is a noble gas, along with helium (He), neon (Ne), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).
- A noble gas, also known as inert gas, is a gaseous element that does not readily combine with other elements and form compounds.
CHARACTERISTICS AND PROPERTIES
- Argon is a colorless, odorless, and tasteless gas at room temperature.
- Even in its liquid and solid form, argon stays colorless.
- When ionized and excited, argon gives off a blue-violet glow.
- Argon is an inert gas at standard temperature and pressure.
- Argon can form certain compounds under extreme conditions.
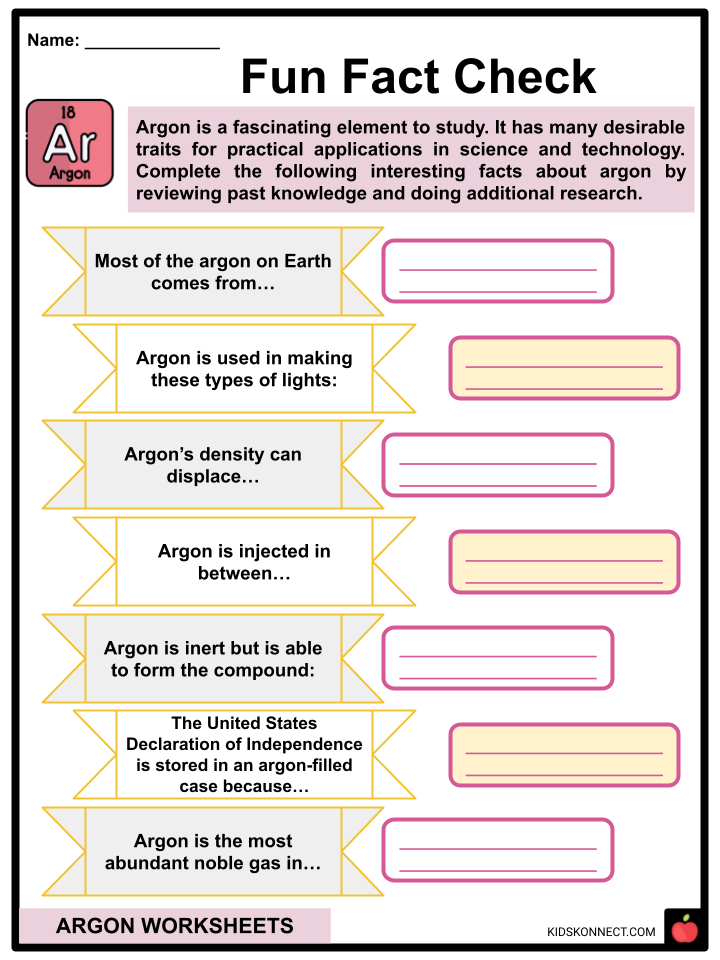
- For example, argon can form a compound with fluorine and hydrogen called argon fluorohydride, which is only stable at very cold temperatures.
- Argon has the same water solubility as oxygen but is more soluble than nitrogen by 2.5 times.
- It is a nonflammable and nontoxic gas, but it poses a risk of asphyxiation because it is 38% more dense than air and may displace oxygenated air in confined spaces.
- Argon is a poor conductor of heat and electricity, making it an ideal material for insulation.
WHERE ARGON IS PRESENT
- Argon is the third-most abundant gas on Earth, following nitrogen and oxygen, excluding water vapor.
- It is the most abundant noble gas.
- Argon comprises about 0.94% of the Earth’s atmosphere.
- Meanwhile, the planet Mercury has 70% argon in its atmosphere.
- Argon atoms are present in air and account for 1% of air’s volume.
- In small trace amounts, argon is present in the Earth’s crust as well as ocean water.
- Usually, argon is synthesized from liquid air as a byproduct of the production of nitrogen and oxygen.
ISOTOPES
- Isotopes are different forms of the same element that have equal numbers of protons but vary in the number of neutrons.
- Argon has three stable isotopes, which are argon-36 (containing 18 neutrons), 38 (containing 20 neutrons), and 40 (containing 22 neutrons).
- Argon-40 is the most abundant isotope, comprising over 99% of argon found on Earth.
- It comes from the radioactive decay of potassium-40.
- Argon-36 is the most common isotope in the universe and is formed by stars in their silicon-burning phase.
- Still, argon is considered rare in the universe.
USES AND APPLICATIONS
- Because it is inert, argon is an ideal material for various industrial processes where an inert environment is required.
- Argon is produced industrially by distilling air cryogenically.
- In the welding industry, it is used as a shield gas.
- In the aerospace industry, it is used as a non-reactive gas in vacuum furnaces.
- In the metal industry, it is used in manufacturing titanium and other reactive metals.
- It is also used to create a blanket atmosphere that is conducive to the growth of silicon and germanium crystals.
- Argon also acts as an insulator in the space between panes in double-paned windows.
- Argon is put into incandescent light bulbs as a protective gas for the filament, as well as fluorescent light bulbs.
- Argon can displace oxygen in a room and stop combustion, making it perfect for fire-suppression systems.
- It can also be used to displace air in food and drinks.
- Pressurized argon is found in aerosol cans as a propellant.
- Liquid argon is useful to destroy cancerous tissue in cryosurgery.
- Argon lasers are used in treating retinal detachment in the eyes.
- Argon is also useful for historical preservation, as it can preserve old documents.
- Argon has no known biological functions.
Argon Worksheets
This fantastic bundle includes everything you need to know about Argon across 25 in-depth pages. These ready-to-use worksheets are perfect for teaching kids about Argon. Argon is found in group 18 of the periodic table of elements, along with helium, neon, argon, krypton, xenon, radon, and oganesson.



Complete List of Included Worksheets
Below is a list of all the worksheets included in this document.
- Argon Facts
- Vocabulary Test
- Arbiters of Argon
- Akin to Argon
- Argon Errors
- Practical Applications
- Industrial Use
- Fun Fact Check
- These Argon!
- Types of Gases
- Argon Infographic
Frequently Asked Questions
What is argon, and where does it come from?
Argon is a chemical element with the symbol “Ar” and atomic number 18. It is a noble gas and is colorless, odorless, and tasteless. Argon is relatively abundant in Earth’s atmosphere, making up about 0.93% of the atmosphere by volume. It is obtained as a byproduct of the fractional distillation of liquid air.
What are the properties and uses of argon?
Argon is an inert gas, which means it is chemically stable and does not readily react with other elements or compounds. Its properties make it useful in various applications, such as:
Welding: Argon is often used as a shielding gas in welding processes to protect the weld area from atmospheric contamination.
Lighting: Argon is used in gas-discharge lamps, including neon signs and fluorescent lighting.
Cooling: In liquid form, argon is used in cryogenic applications to achieve extremely low temperatures.
Scientific research: Argon is used as a carrier gas in gas chromatography and as a detector gas in nuclear physics experiments.
What is the role of argon in Earth’s atmosphere?
Argon is a minor component of Earth’s atmosphere but plays a crucial role in radiometric dating. It is used in the potassium-argon dating method to determine the age of rocks and minerals. This method relies on the radioactive decay of potassium-40 to argon-40.
Is argon dangerous to humans?
Argon is generally not considered dangerous to humans in normal atmospheric concentrations. It is non-toxic and non-flammable. However, in high concentrations, argon can displace oxygen and pose an asphyxiation risk in enclosed spaces. Proper safety measures should be followed when working with argon in confined areas.
How is argon produced for industrial use?
Argon is typically produced as a byproduct of the air separation process. This process involves cooling and compressing air to separate it into its individual components, including nitrogen, oxygen, and argon. The argon is then purified through further cryogenic distillation to obtain high-purity argon gas for industrial applications.
Link/cite this page
If you reference any of the content on this page on your own website, please use the code below to cite this page as the original source.
Link will appear as Argon Facts & Worksheets: https://kidskonnect.com - KidsKonnect, November 8, 2023
Use With Any Curriculum
These worksheets have been specifically designed for use with any international curriculum. You can use these worksheets as-is, or edit them using Google Slides to make them more specific to your own student ability levels and curriculum standards.



