Download This Sample
This sample is exclusively for KidsKonnect members!
To download this worksheet, click the button below to signup for free (it only takes a minute) and you'll be brought right back to this page to start the download!
Sign Me Up
Table of Contents
Boron is a chemical element classified as a metalloid. A metalloid has properties of both metals and nonmetals. In the periodic table of elements, it is the first one in the thirteenth column, represented by the symbol B, and has an atomic number of 5. In its pure crystalline form, boron is black, brittle, and lustrous. It can serve as a semiconductor and is utilized in fertilizers, insecticides, detergents, cosmetics, insulation, flame retardants, and antiseptics, among other applications.
See the fact file below for more information about Boron, or you can download our 28-page Boron worksheet pack to utilize within the classroom or home environment.
Key Facts & Information
ETYMOLOGY
- The name boron comes from the Arabic word “buraq,” the name for the mineral borax.
HISTORY
- Boron has been discovered on Earth in rocks that are over 3.8 billion years old and meteorites that have fallen from Mars to Earth.
- The most common source of boron on Earth is borax.
- Borax had been used for thousands of years before it was even discovered as an element.
- In 300 ADE, borax was a material for ceramic glazes in China.
- It was also exported as crystallized deposits from lake beds in Tibet in the 8th century ADE and used as a flux by goldsmiths and silversmiths.
- Boron was discovered and extracted as an element in 1808, simultaneously by English chemist Sir Humphry Davy and French chemists Joseph L. Gay-Lussac and Louis J. Thenard.
- Davy produced boron by heating borax with potassium.
- He called the newly discovered element boracium.
- Meanwhile, Gay-Lussac and Thenard used iron instead of potassium.
- Their efforts, however, did not result in producing pure boron.
- Pure boron was reportedly produced a century later, in 1909, by American chemist Ezekiel Weintraub.
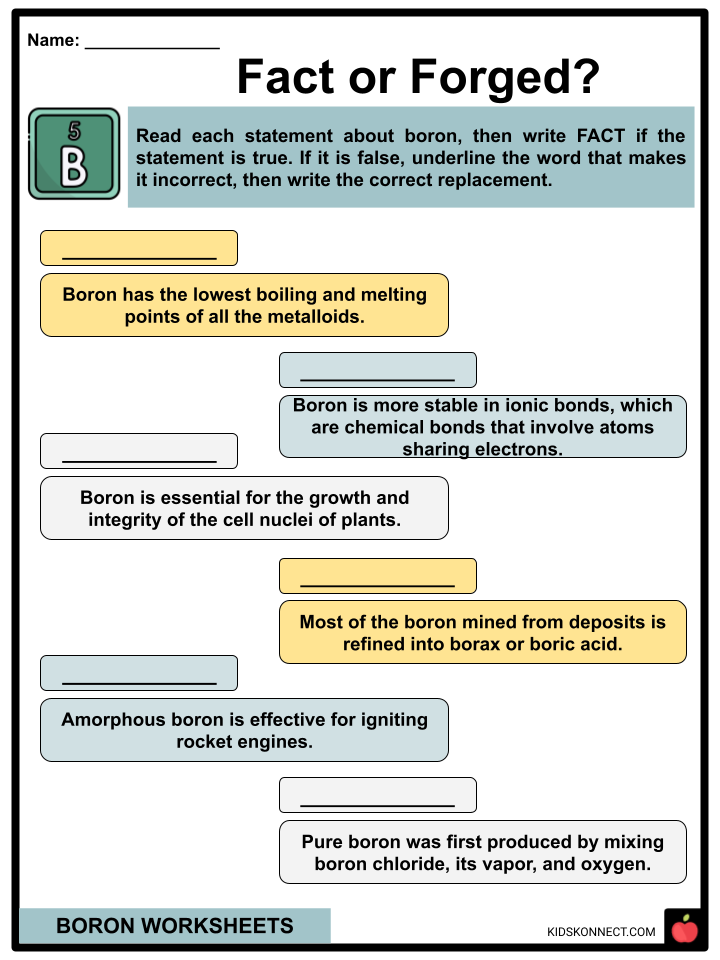
- Weintraub obtained the pure element by mixing boron chloride, its vapor, and hydrogen.
ELEMENT QUICK FACTS
- In the periodic table, boron is represented by the symbol “B” and the atomic number 5.
- Boron has 5 protons, 5 electrons, and 6 neutrons.
- Boron belongs to group 13, which is also called the boron group.
- It is part of period 2 (the second row) and the p-block of the periodic table.
- The atomic weight of boron is 10.811 u.
- Its boiling point is 3,927°C, and its melting point is 2,076°C.
- At room temperature, boron is in a solid state.
- Boron has the highest boiling and melting points of all the metalloids in the periodic table of elements.
- The density of boron is 2.37 grams per cubic centimeter.
- Boron is more stable in covalent bonds, the chemical bond that involves atoms forming electron pairs by sharing electrons.
- It does not usually form bonds by one atom losing an electron and gaining them from another, which is called an ionic bond.
- It is difficult for borons to form ionic bonds because it is extremely small and has a high charge density.
MORE CHARACTERISTICS
- Crystalline boron is lustrous and has a dark color: either black, brown, or silvery.
- It is unreactive in a crystalline form but can be reactive as an amorphous powder.
- Boron is generally not toxic, but large amounts can upset the body’s metabolism.
- Crystalline boron is considered the second-hardest element, following the diamond, hitting a 9.3 on the Mohs scale.
- Though solid, it may be too brittle to be used in tools.
- It is a good semiconductor, which means it conducts heat and electricity at high temperatures and can serve as an insulator at low temperatures.
- Boron is considered more stable than carbon.
- One way to identify boron is to put it through a flame test; if the substance burns bright green, then it’s boron.
ISOTOPES AND ALLOTROPES
- There is said to be 13 isotopes of boron.
- An isotope is a variation of an element that has the same atomic number but a different atomic mass of such an element.
- Boron has only two stable isotopes that are naturally occurring: Boron-10 (which accounts for 19.9% of natural boron) and Boron-11 (80.1% in abundance).
- Boron-10 contains five neutrons, while Boron-11 contains six.
- Boron exists in four major allotropes, which are different structural forms of the same element.
OCCURRENCE
- Boron is a rare element on Earth and even in the whole universe.
- It does not occur naturally as a pure element.
- It is usually found in compounds, such as borax and boric acid, mined from sedimentary rock formations and deposits in the Earth’s crust.
- Turkey is the country with the biggest deposits of boron in the world.
- Approximately 72% of the world’s known deposits of boron are found in Turkey.
- The second-largest producer of boron is the United States of America.
- Argentina, China, Russia, and Chile also have a large number of boron reserves.
- Kernite, the most important source of boron, also known as rasorite, is found in the Mojave Desert in California, USA.
BIOLOGICAL ROLE
- Boron contributes significantly to plant growth, particularly to the cell walls of plants.
- Small doses of boron are not poisonous to animals.
USES AND APPLICATIONS
- Because of its bright green flame, boron is used to manufacture fireworks and other components in pyrotechnics.
- Boron is also used as an additive to steel to increase its hardness.
- Boron can also be used to increase the resistance of glass to heat shock.
- This is why some laboratory glassware and cookware materials are made of borosilicate glass.
- Amorphous boron is effective for igniting rocket engines.
- The boron-10 isotope is a neutron absorber, which makes it an ideal material in nuclear reactors to control the fission rate.
- The boron-11 isotope is used in manufacturing nuclear weapons and in medical imaging.
- Boron fibers are used as structural materials in aircraft and spacecraft.
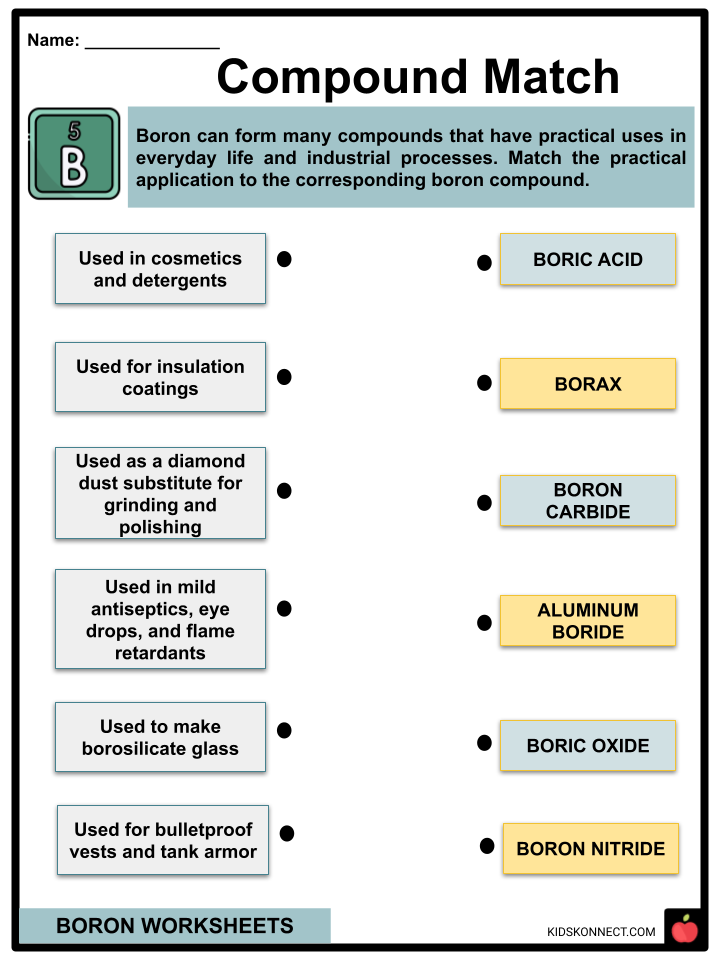
- Most of the boron mined from deposits is refined into borax or boric acid.
- Borax, which has been used for thousands of years, is commonly used in the present day as an ingredient in cleaning products and detergents.
- Borax is also used in cosmetics and tile glazes.
- Boric acid is an effective insecticide against cockroaches and fleas.
- Boric acid is also used in mild antiseptics, eye drops, and flame retardants or materials that slow the spread of fire.
- Boron carbide is an effective material for bulletproof vests and tank armor.
- Aluminum boride can be used as a diamond dust substitute for grinding and polishing.
Boron Worksheets
This fantastic bundle includes everything you need to know about Boron across 28 in-depth pages. These ready-to-use worksheets are perfect for teaching kids about Boron. In its pure crystalline form, boron is black, brittle, and lustrous. It can serve as a semiconductor and is utilized in fertilizers, insecticides, detergents, cosmetics, insulation, flame retardants, and antiseptics, among other applications.


Complete List of Included Worksheets
Below is a list of all the worksheets included in this document.
- Boron Facts
- Boron Basics
- Guess the Word
- History in a Nutshell
- Metalloid Spotting
- Compound Match
- Multipurpose Element
- Fact or Forged?
- Map of Abundance
- Fun Fact Acrostic
- In My Own Words
Frequently Asked Questions
What is boron?
Boron is a chemical element with the symbol “B” and atomic number 5. It is a metalloid, which means it has properties of both metals and nonmetals. Boron is found in nature primarily in the form of borates, which are compounds containing boron, oxygen, and other elements.
Where is boron commonly found?
Boron is commonly found in nature in the form of borate minerals, such as borax, colemanite, and ulexite. These minerals are often mined in arid regions, including the southwestern United States, Turkey, and Chile. Boron is also present in small amounts in soil and some groundwater.
What are the uses of boron?
Boron has several important uses. It is used in the manufacture of glass and ceramics, where it can improve their thermal and mechanical properties. Boron is also used in the production of boron steel, which is known for its high strength and durability. Additionally, boron compounds are used in agriculture as micronutrients for plants, and boron neutron capture therapy (BNCT) is a medical application for cancer treatment.
What are some interesting properties of boron?
Boron has some unique properties, including its ability to absorb neutrons, which makes it useful in nuclear applications. It is also known for its high melting point and low density. Boron compounds can fluoresce under certain conditions, and boron has a wide range of stable isotopes.
Is boron safe for humans and the environment?
Boron is generally safe for humans when used in moderation. It is an essential micronutrient for plants, but excessive exposure to boron can be harmful. Boron compounds are used in pesticides, so care must be taken to use them according to safety guidelines. Boron is not considered highly toxic to humans, but ingestion of large amounts can lead to health problems. In the environment, excessive boron levels can be harmful to aquatic life, so there are regulations in place to limit boron discharges into water bodies.
Link/cite this page
If you reference any of the content on this page on your own website, please use the code below to cite this page as the original source.
Link will appear as Boron Facts & Worksheets: https://kidskonnect.com - KidsKonnect, October 19, 2023
Use With Any Curriculum
These worksheets have been specifically designed for use with any international curriculum. You can use these worksheets as-is, or edit them using Google Slides to make them more specific to your own student ability levels and curriculum standards.



