Download This Sample
This sample is exclusively for KidsKonnect members!
To download this worksheet, click the button below to signup for free (it only takes a minute) and you'll be brought right back to this page to start the download!
Sign Me Up
Table of Contents
Helium is the second most abundant element in the Earth’s atmosphere, next to hydrogen. It is a noble gas, which means it does not typically mix with other chemicals and form new compounds. It is colorless, odorless, and tasteless. Among all the elements in the periodic table, helium has the lowest boiling point. A common use for helium gas is for the inflation of balloons because of its low density. Another application for helium is when welders use it as a shielding gas. It is also used to pressure liquid-fueled rocket fuel tanks.
See the fact file below for more information about Helium, or you can download our 27-page Helium worksheet pack to utilize within the classroom or home environment.
Key Facts & Information
ETYMOLOGY
- The name helium is derived from the Greek word “helios,” which means sun.
- Helios is also the name of the Greek god of the sun.
- Before it was discovered on Earth, helium had already been detected surrounding the sun.
- The name helios was given by English astronomer Norman Lockyer and English chemist Edward Frankland.
HISTORY
- The person who first detected helium in the gaseous atmosphere around the sun was French astronomer Pierre Jules C. Janssen.
- Janssen traveled to India in 1868 to observe the solar spectrum during a total eclipse.
- While observing through a prism, he saw a bright yellow line in the spectrum of the Sun’s chromosphere.
- This line indicated a new element, but Janssen initially thought it represented sodium.
- That same year, an English astronomer named Joseph Norman Lockyer observed the sun through London smog and noticed the same yellow spectral line.
- Lockyer came to the conclusion that it was not sodium; instead, it was an element in the Sun yet to be known on Earth.
- In naming the unknown element, Lockyer and chemist Edward Frankland decided to take inspiration from the Greek word for sun.
- Helium was the first element not to be discovered on Earth.
- In 1882, Italian physicist Luigi Palmieri was analyzing the gas emissions from Mount Vesuvius when he identified the same yellow line.
- In 1889, the yellow line also appeared in the gas emission of the mineral uraninite (UO2) as it dissolves in acid — an observation made by American geologist William Hillebrand.
- It was in 1895 when the element was discovered on Earth and conclusively identified by the British chemist Sir William Ramsay.
- Eight years later, in 1903, Ramsay also teamed up with Frederick Soddy to determine that helium is a product of the spontaneous disintegration of radioactive substances.
- In 1895, helium’s atomic weight was measured by Per Teodor Cleve and Nils Abraham Langer at Uppsala, Sweden.
ELEMENT QUICK FACTS
- Helium is classified as a noble gas, just like argon, neon, krypton, xenon, and radon, which are gases that are largely unreactive to other elements or compounds.
- Noble gases have full outer electron shells, which means it does not share, give, or accept electrons.
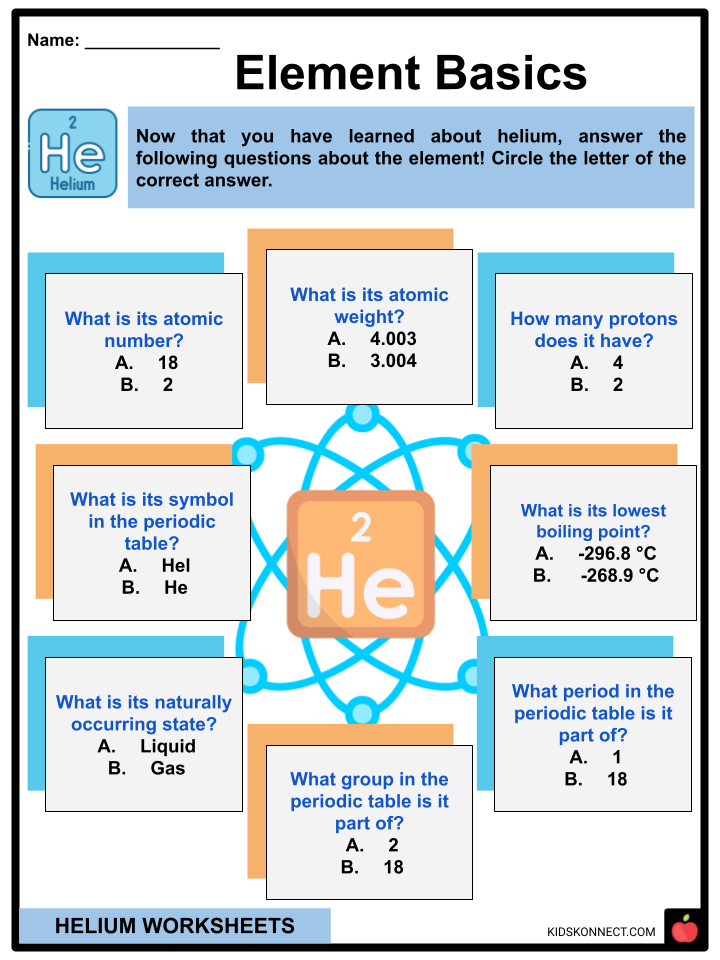
- Helium has an atomic number of 2, which means each atom has two protons.
- It is represented by the symbol He in the periodic table.
- It is the first element in the 18th and last column of the periodic table; therefore, it’s part of group 18 and period 1.
- Helium atoms have 2 electrons and 2 protons.
- Its atomic weight is 4.003, which is also seen in the periodic table.
- Next to hydrogen, helium is the second lightest element, with a density of 0.000164 grams per centimeter cubed at 20 degrees Celsius.
- Helium condenses to a liquid at temperatures close to absolute zero.
- Helium has the lowest boiling point of all elements, which is -268.9 degrees Celsius.
- It is the only element that will not turn into a solid state by cooling at normal atmospheric pressure.
ABUNDANCE
- Helium is the second-most abundant element in the universe.
- It makes up approximately 23% of the mass of the universe.
- In stars, helium is produced after hydrogen atoms smash together by nuclear fusion.
- At the Sun’s core, 695 million tons of helium gas are converted from hydrogen every second.
- Helium is rare on Earth and is often found with natural gas in the Earth’s crust.
- There is only 0.000524% of helium in the Earth’s atmosphere because its atoms are so light that they escape into space. The Earth’s gravity is not strong enough to keep it grounded.
- It is more abundant underground, where there are large natural gas deposits.
- Helium is also formed from alpha-particle decay of radioactive elements such as uranium and thorium.
- It is considered a nonrenewable resource on Earth.
CHARACTERISTICS
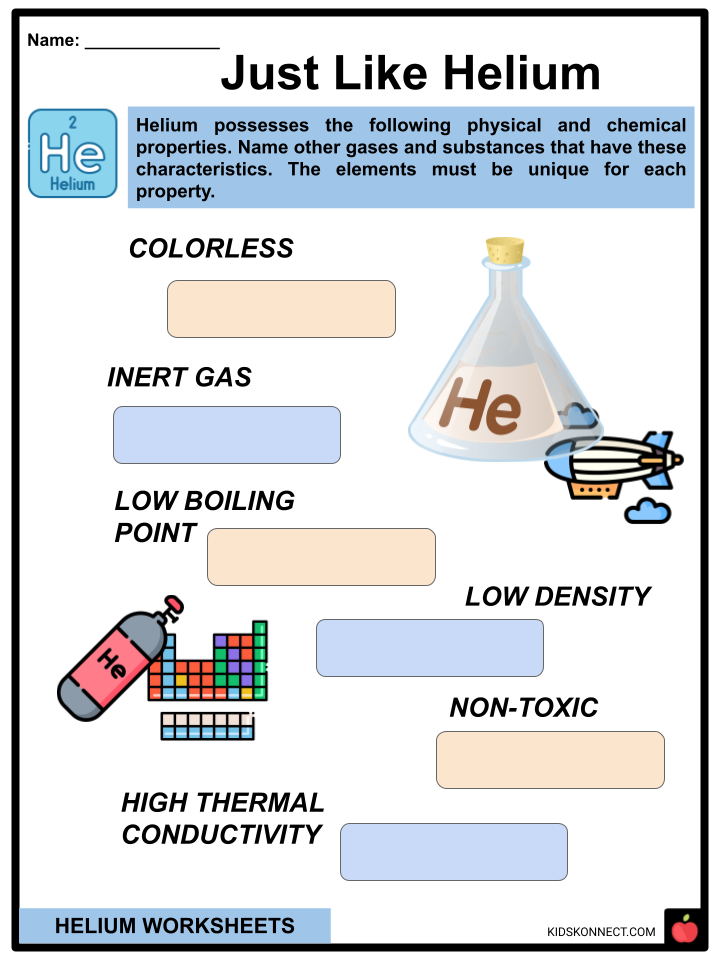
- Helium is a colorless, tasteless, odorless, non-toxic, and unreactive noble gas.
- Helium’s naturally occurring state is gaseous.
- At absolute zero, it is liquefied.
- For helium to be turned into a solid state, it must be pressurized.
- Because of its non-reactivity, it is the most stable of all the elements.
- It does not form compounds or burn with other elements under standard conditions.
- Following neon, helium is the second-least reactive noble gas.
- Helium is monatomic, which means it is found as single atoms of the element.
USES AND EFFECTS
- Helium is often used in inflating party balloons because of its low density; the gas is stored in cylinders synthesized by radioactive decay of uranium and thorium.
- It is also used to fill weather balloons and airships.
- When someone inhales helium, their voice temporarily changes, seemingly increasing the pitch, but it actually does not.
- Helium is non-toxic, but breathing it can lead to oxygen deprivation and asphyxiation, depending on the situation.
- It is also used as a cooling medium for superconducting magnets found in NMR spectrometers and MRI scanning machines.
- Helium is an ideal cooling medium for satellite instruments and the liquid oxygen and hydrogen that powered the Apollo spacecraft.
- Helium is also used to supply a protective, inert atmosphere conducive to chemical storage, fiber optics, and semiconductor manufacturing.
- It is used to create an inert gas shield in arc welding.
- Helium is used to inflate car airbags upon impact and detect leaks in car air-conditioning systems.
- It is also used to test seals of aerosol products, tires, air conditioners, refrigerators, and fire extinguishers.
- Helium-neon gas lasers are used in eye surgery and barcode scanners.
Helium Worksheets
This fantastic bundle includes everything you need to know about Helium across 27 in-depth pages. These ready-to-use worksheets are perfect for teaching kids about Helium. Helium is the second most abundant element in the Earth’s atmosphere, next to hydrogen. It is a noble gas, which means it does not typically mix with other chemicals and form new compounds.




Complete List of Included Worksheets
Below is a list of all the worksheets included in this document.
- Helium Facts
- Expound and Illustrate
- Element Basics
- Picture Clues
- Truth Balloons
- Helium History
- Discoverer Match
- Noble Gas Search
- Just Like Helium
- Abundant Elements
- Science In A Song
Frequently Asked Questions
What is helium?
Helium is a chemical element with the atomic number 2 and the symbol He. It is a colorless, odorless, and tasteless noble gas that is the second-lightest and second-most abundant element in the universe.
Where is helium found?
Helium is primarily extracted from natural gas deposits, where it is typically found as a trace element. It is produced as a byproduct of natural gas processing, and the United States is one of the largest producers of helium in the world.
What are the main uses of helium?
Helium has several important applications, including: a. Inflating balloons and airships due to its low density and non-flammable nature. b. Cooling in scientific research, such as in superconducting magnets and cryogenics. c. Leak detection in various industries because helium atoms are small and can penetrate tiny openings. d. Welding, as it can shield metals from contamination and reduce heat distortion. e. Medical applications, like MRI machines where it is used to cool the magnets.
Why is helium important?
Helium is essential for various scientific, industrial, and medical purposes. Its unique properties, such as its low boiling point and inertness, make it irreplaceable in certain applications, particularly in cooling superconducting materials and conducting research in low-temperature physics.
Is there a helium shortage?
Helium availability has been a concern in the past due to its limited natural sources and the overuse of the helium stored in the U.S. National Helium Reserve. While there have been periods of helium shortages, efforts to conserve helium and explore new sources have helped mitigate these issues. However, helium remains a finite resource, and it’s important to use it responsibly.
Link/cite this page
If you reference any of the content on this page on your own website, please use the code below to cite this page as the original source.
Link will appear as Helium Facts & Worksheets: https://kidskonnect.com - KidsKonnect, November 8, 2023
Use With Any Curriculum
These worksheets have been specifically designed for use with any international curriculum. You can use these worksheets as-is, or edit them using Google Slides to make them more specific to your own student ability levels and curriculum standards.



